Tag: Pharmaceutical R&D
-

After Approval: Running Safety, Real World Evidence, and the Product Lifecycle
This is part 7 and the last part of The Drug Development Playbook. Post‑approval is less about molecules and more about systems, signals, and evidence strategy.
-

From Pivotal Data to Approval: The Regulatory Review Strategy
This is Part 6 of The Drug Development Playbook. This article explains how to turn that data and your manufacturing story into an approval.
-
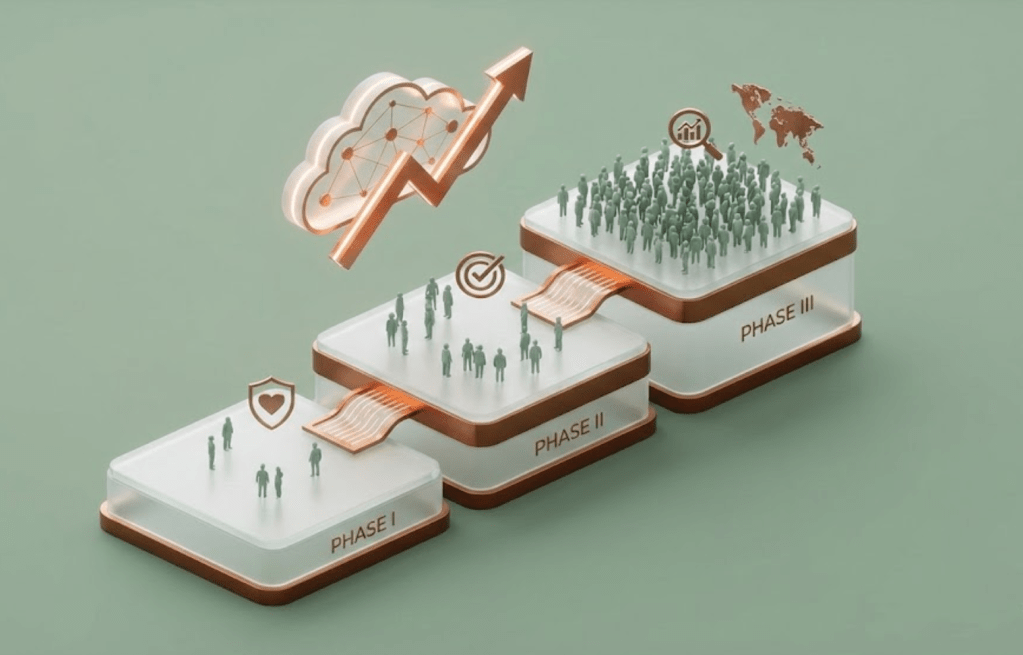
Clinical Development: Phases I to III
This is Part 5 in The Drug Development Playbook. In this article we move into clinical trials from phase I to III.
-

Preclinical Development and IND/CTA Readiness: making the candidate safe enough to test in people
This is Part 4 of The Drug Development Playbook. We talk about building nonclinical and CMC evidence that lets regulators say yes to first-in-human trials.
-

How Genomics Is Changing Drug Target Selection in Big Pharma
The integration of human genetics in pharmaceutical research significantly enhances drug development. By prioritizing targets with genetic support, companies can improve trial efficacy and reduce risks. Utilizing genomics allows for better indication selection, safety predictions, and asset valuation. This strategic approach fosters innovation while addressing ethical considerations in clinical trials.