Tag: biotech strategy
-

After Approval: Running Safety, Real World Evidence, and the Product Lifecycle
This is part 7 and the last part of The Drug Development Playbook. Post‑approval is less about molecules and more about systems, signals, and evidence strategy.
-

From Pivotal Data to Approval: The Regulatory Review Strategy
This is Part 6 of The Drug Development Playbook. This article explains how to turn that data and your manufacturing story into an approval.
-
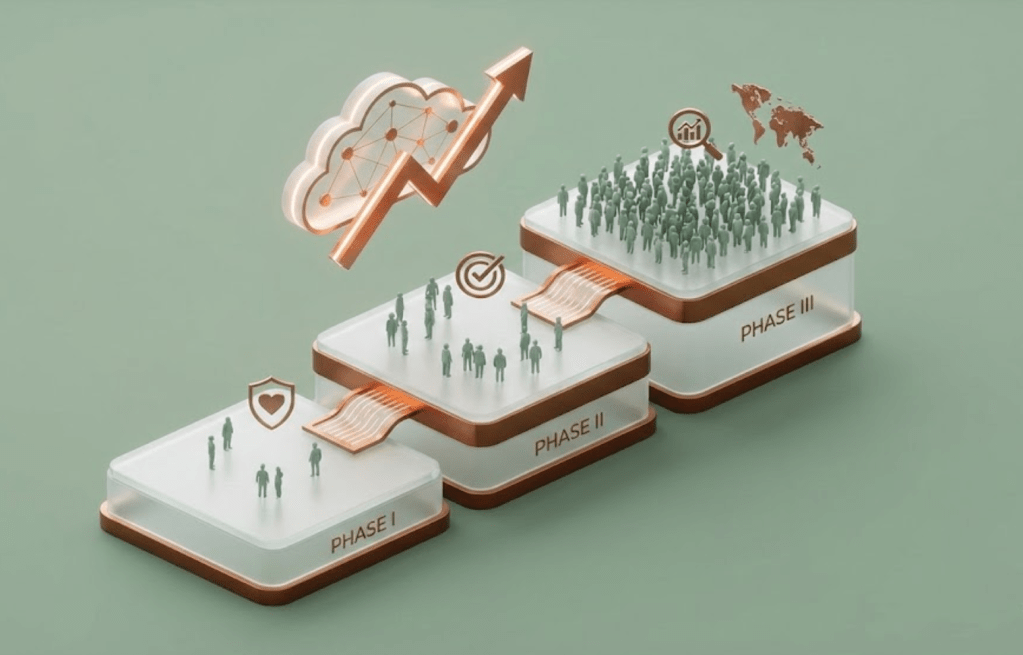
Clinical Development: Phases I to III
This is Part 5 in The Drug Development Playbook. In this article we move into clinical trials from phase I to III.
-

Preclinical Development and IND/CTA Readiness: making the candidate safe enough to test in people
This is Part 4 of The Drug Development Playbook. We talk about building nonclinical and CMC evidence that lets regulators say yes to first-in-human trials.
-

Disease Understanding and Target Identification: Picking the Biology Worth Backing
This is Part 2 of The Drug Development Playbook, an eight-part series on how medicines actually move from idea to patient use and how smarter decisions at each stage change outcomes.
-

From Idea to Patient: How Drug Development Actually Works
This is the first article in The Drug Development Playbook, an eight-part series on bringing a medicine from idea to patient use. Part 1 outlines the full journey, key stages, and where most programs fail, setting up practical deep dives into each phase that follow in the rest of the series.