Author: Aryan Kenia
Life Sciences Analyst
I remember the exact afternoon in the lab during my MSci at University College London when a promising assay gave us a clean, exciting result. We ran the data, celebrated quietly, and then someone asked the uncomfortable question: “Can we make ten thousand doses of this safely and cheaply if it works?” The room grew quiet. That question stuck with me through internships and applied R&D work, and it shaped how I now look at targets and technologies.
Breakthrough biology wins headlines. But for patients and payers, the real test arrives when you try to make treatments at scale, consistently, and affordably. This article explains why scalability matters more than a single scientific win, how companies should evaluate scale early, and what practical changes R&D and commercial teams should make.
Why scalability should be your top strategic filter
A novel mechanism can excite investors and scientists. Yet many promising discoveries stall because teams underestimate manufacturing complexity, regulatory expectations, or cost-of-goods hurdles. Science proves a concept. Scalability proves a product.
When you plan projects with scale in mind, you force clearer choices about modality, supply chain, and commercial strategy. You also reduce the chance of hitting a cliff at Phase 3 or at launch when demand, regulation, or cost becomes unforgiving.
A practical example from early-career work
At an internship, I ran feasibility assays that showed good analytic separation and stable batches at lab scale. The biology looked clean. But when I compared our lab process to what a contract manufacturer would require at 1,000-litre scale, the gap looked large. The raw materials had long lead times. The assay required a reagent that sits outside standard GMP suppliers. We paused and redesigned several steps. That early course correction saved the project from a late-stage scramble and taught me a practical rule: plan for scale before you commit full clinical spend.
The four scalability axes that decide commercial fate
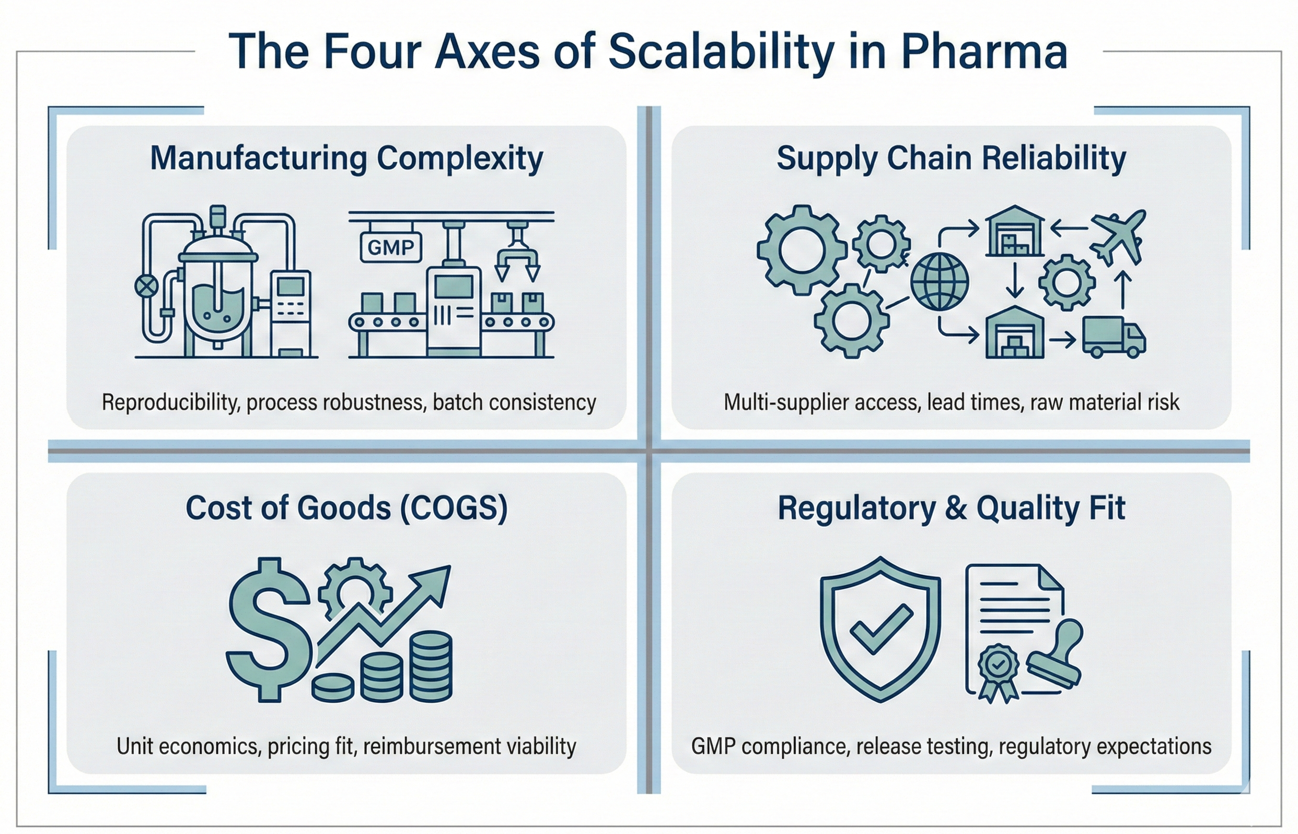
Think of scalability as four axes you must score before you commit major resources.
- Manufacturing complexity and reproducibility
Can you move from bench protocols to GMP lines without custom equipment or fragile steps? If a process relies on unstable intermediates or bespoke materials, you face higher failure risk and cost. - Supply chain reliability
Are raw materials available from multiple qualified suppliers? Do any inputs require long lead times or single-source suppliers? Fragile supply chains amplify risk and can halt production. - Cost-of-goods (COGS) and pricing fit
Even if a drug works, can payers afford it? Therapies with astronomical COGS need very strong clinical benefit and a clear reimbursement path. Early COGS estimates feed commercial planning. - Regulatory and quality expectations
Regulators expect consistent manufacturing and control strategies. If your process introduces variability, you increase the chance of extended inspections, delays, or additional studies.
Score each axis honestly. Many projects fail on one weak axis rather than failing across all four.
How scalability changes modality choice
Modality matters. Small molecules, monoclonal antibodies, oligonucleotides, cell therapies, and viral vectors each come with distinct scale profiles.
- Small molecules often scale well on established chemistries and contract manufacturing networks.
- Antibodies require biologics manufacturing know-how and cold chain logistics.
- Cell and gene therapies face steep manufacturing, logistics, and patient-specific constraints that challenge large-scale rollout.
Match the biology to a modality that fits a plausible commercial model. If biology suggests a target that only cell therapy can address, make sure the clinical and commercial case justifies the extra manufacturing complexity.
Quick checklist to assess scalability at project start
You can run a short, repeatable assessment that keeps teams honest. Use this checklist before advancing beyond lead optimisation:
- Can a 1,000-litre manufacturing recipe exist for this process?
- Are there at least two GMP suppliers for critical raw materials?
- Does estimated COGS allow a realistic pricing band with expected payer pressure?
- Are critical quality attributes measurable with standard release assays?
- Have we scoped the regulatory data package needed to demonstrate consistent manufacture?
If the answer is “no” on two or more items, pause and redesign. Early iteration beats late rescue.
Organizational moves that actually create scale readiness
Big Pharma and consultancies often talk about scale readiness without changing processes. Practical changes work better.
- Embed process and CMC expertise in target teams
Put a manufacturing or CMC representative on early project teams. Don’t relegate scale questions to a later gate. Early presence forces realistic design constraints. - Require a “scale impact” score at portfolio gates
Add a short, standard scorecard that flags manufacturing complexity, supply fragility, and COGS risk. Make decisions with the score visible. - Build supplier and CMOs relationships early
Talk to contract manufacturers and raw material suppliers during lead optimisation. Their feedback narrows technical risk and timelines. - Model commercial scenarios with realistic COGS
Run a simple model that ties price, uptake, and COGS to revenue and margin. If the math doesn’t work, change modality or target selection.
These changes move scale from an abstract worry to an actionable constraint.
Regulatory and payer realities you cannot ignore
Regulators now expect consistent manufacturing and pharmacovigilance plans. Payers ask about value relative to cost. If a therapy depends on complex, single-batch manufacturing, payers will press on comparative benefit and budget impact. Answer these questions early. Don’t wait for the regulator or payer to force trade-offs during registration or reimbursement negotiations.
Common traps I see teams fall into
- Chasing novelty without industrial fit. Scientists chase elegant mechanisms, but the industry needs reliable products.
- Underestimating supply chain fragility. A single critical reagent with a 6-month lead time creates program risk.
- Treating CMC as an afterthought. People assume “we’ll fix manufacturing later” and then face massive redesign.
- Ignoring patient access. A treatment that costs more than the system will tolerate will face slow uptake even when clinically strong.
Short case illustration: when scale beats novelty
Take a small team that has a clever biologic with a novel adjuvant. The science promises high potency, but the adjuvant requires a rare lipid ingredient with single-source manufacture. Early conversations with potential CMOs and suppliers reveal multi-month waits and complex qualification. The team pivots to a different formulation that gives slightly lower potency in vitro but uses standard, multi-supplier materials. The pivot lets them keep timelines, reduce COGS, and ultimately enables a viable commercial launch scenario.
You want to be the team that makes this pragmatic choice early, not the team that redesigns at Phase 3.
Key takeaways
- Score scalability early on manufacturing complexity, supply reliability, COGS, and regulatory fit.
- Align modality choice to industrial realities, not only to the biological ideal.
- Embed CMC expertise and supplier feedback at the start to avoid expensive late pivots.

Leave a comment