Author: Aryan Kenia
Life Sciences Analyst
I still remember sitting over notes from my MSci project and thinking: if we can point to a human gene and say, with real data, that it affects disease risk, why would we pick targets any other way? That question has guided my work since my time at University College London and through internships where I moved between the bench and commercial teams. Genomics gives you human evidence early, and that changes how teams pick targets, design trials, and price risk.
Below I explain the practical changes I see in big pharma, show real examples, and share concrete steps R&D leaders should adopt if they want better odds in development.
Why human genetics matters for target selection
Pharma teams used to rely mostly on animal models, cell assays, and intuition. Those still matter, but they often mislead because human biology differs from model systems. Genomics gives you direct information about how genetic variation in people relates to disease. That signal tells you whether altering a protein’s function in humans likely moves the needle on disease risk.
Researchers have shown that drug programs built around targets with human genetic support increase their chance of approval. That finding forces a simple shift: prioritize targets with human evidence, then layer in functional work to understand how to act on them.
A clear example: PCSK9
The PCSK9 story shows how human genetics can point teams to a target worth investing in. Researchers found people with rare loss-of-function variants had much lower LDL and fewer cardiovascular events. That human observation gave pharma a real downstream readout to aim for. Companies developed PCSK9 inhibitors and produced drugs that actually lower events in trials. That pathway from human variant to approved drug provides a repeatable template for other programs.
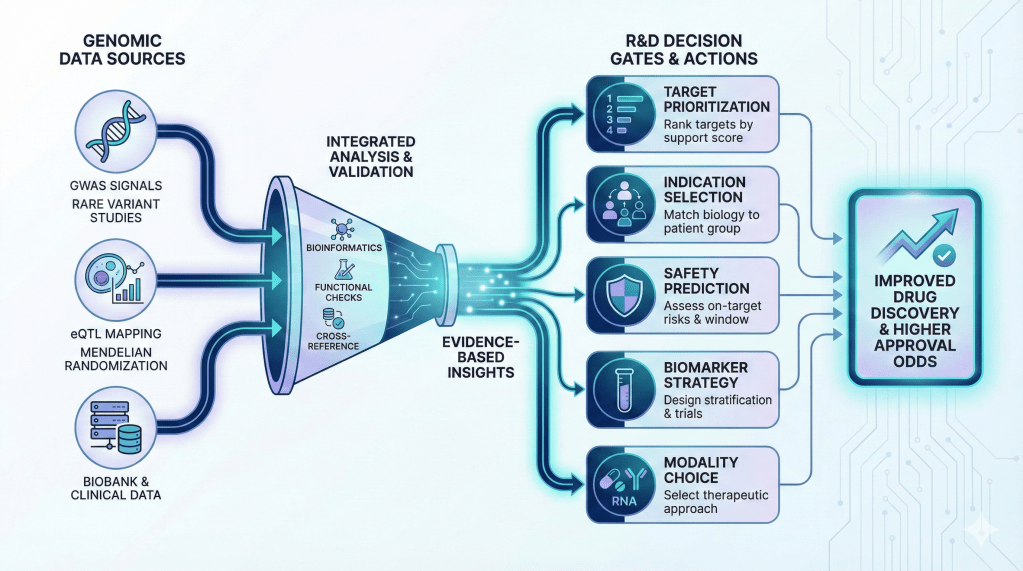
Five practical ways teams use genomics right now
1) Prioritise targets with genetic support
When a GWAS signal or rare-variant study implicates a gene, teams move that target up the list. Genetic evidence does not guarantee success, but it raises the odds. Use genetic evidence as a ranking tool during portfolio reviews.
2) Pick the right indication
Genomics helps you match biology to the patient group most likely to benefit. A gene might affect disease only in a subgroup. Use Mendelian randomization and interaction analyses to refine indication choice before you commit to expensive trials.
3) Predict on-target safety and the therapeutic window
Human loss-of-function or gain-of-function variants act like natural trials. If people tolerate loss of function across their lifespan, you gain confidence an inhibitor might avoid severe safety signals. If variants cause developmental issues, plan for monitoring or avoid the mechanism.
4) Design biomarkers and stratification strategies
Genetic markers can become predictive biomarkers or population stratifiers. Use them to run smaller, more efficient trials by increasing the trial’s signal-to-noise ratio.
5) Shape M&A and due diligence
When business development evaluates an asset, human genetic evidence affects valuation and negotiation. If genetics supports the target, buyers pay more and assume less scientific risk.
Data partnerships, access, and commercial strategy
Big Pharma wins when it partners with biobanks, academic consortia, and data platforms. Secure data access, negotiate clear data-use terms, and build pipelines that convert raw genotype-phenotype links into commercial insight. For business development, that means you can price assets with more certainty and avoid surprise risks during late-stage due diligence.
Tools that add confidence (and what they tell you)
Use GWAS to find common-variant signals, and rare-variant sequencing to find genes with large effects. Use eQTL mapping to connect variants to gene expression. Use Mendelian randomization to test causal direction. Treat these tools as complementary. If multiple lines point to the same gene, your confidence grows. If signals disagree, dig into mechanism and cohort differences.
Where the genetics signal affects modality choice
Different modalities answer different biology. Small molecules, antibodies, RNA therapeutics, and cell therapies each carry different risks and manufacturing hurdles. Genetics helps you choose the right modality by clarifying mechanism. If human variants suggest loss of function improves outcome, consider inhibitors or antisense oligonucleotides. If a variant causes gain of function, think about blocking antibodies or targeted degradation. Making this choice early saves time and capital.
How to run a quick genetics sanity check for a program
You do not need a full bioinformatics team to ask the right first questions. Run a three-step sanity check.
- Search for GWAS hits near the gene.
- Look for rare loss-of-function variants in public databases.
- Run a quick Mendelian randomization summary if a GWAS links a biomarker to disease.
If two of three steps show supporting evidence, assign the program a stronger genetic score at the next portfolio gate. That small, repeatable process makes genetic signals actionable.
What I learned from my Human Genetics degree at University College London
As I reviewed the literature on statin-associated muscle symptoms during a UCL project, I saw how genetic evidence clarified mechanisms behind adverse events. Genetic signals helped separate plausible on-target biology from coincidental associations. That experience taught me to treat human genetics as a strategic filter, not a final verdict.
How to present genetic evidence to non-geneticists
You need a short, consistent briefing format for leadership. I use three slides.
- Slide 1: Evidence summary. One table that shows GWAS hits, rare variants, and MR results.
- Slide 2: Mechanism and modality. How the gene links to biology and which therapy types match it.
- Slide 3: Risk and next steps. Safety flags, biomarker candidates, and an immediate experiment plan.
Ethical, regulatory, and patient considerations
Genomics raises ethics and privacy questions that affect trial design and recruitment. You must plan for consent that covers genetics, think about return-of-results policies, and ensure patient privacy when sharing genotype-linked data. Regulators will ask for clarity on how genetic stratification affects generalisability. Answer those questions early and you reduce friction downstream.
Common mistakes teams make
First, teams treat genetic evidence like a pass/fail test. It rarely works that way. Genetics shows degrees of support. Pair it with functional validation. Second, teams assume effect sizes from lifetime genetic exposure translate directly to short clinical trials. They do not. Make explicit pharmacology and trial design plans to bridge that gap.
Practical steps R&D leaders should adopt now
- Embed genetics expertise inside drug teams. External reports help, but in-house experts speed decisions.
- Require a genetic evidence summary at portfolio gates. Put a simple genetic score next to safety and efficacy metrics.
- Secure access to large, harmonised biobanks and clinical datasets. Better data improves the signal.
- Add a three-slide briefing template for pipeline reviews and ask for it at the next gate.
References
- Nelson, M. R., Tipney, H., Painter, J. L., Shen, J., Nicoletti, P., Shen, Y., Floratos, A., Sham, P. C., Li, M. J., Wang, J., Cardon, L. R., Whittaker, J. C., & Sanseau, P. (2015). The support of human genetic evidence for approved drug indications. Nature genetics, 47(8), 856–860. https://doi.org/10.1038/ng.3314
- Jaworski, K., Jankowski, P., & Kosior, D. A. (2017). PCSK9 inhibitors – from discovery of a single mutation to a groundbreaking therapy of lipid disorders in one decade. Archives of medical science : AMS, 13(4), 914–929. https://doi.org/10.5114/aoms.2017.65239
- Chen, R., Duffy, Á., & Do, R. (2025). Genomics of drug target prioritization for complex diseases. Nature reviews. Genetics, 10.1038/s41576-025-00904-4. Advance online publication. https://doi.org/10.1038/s41576-025-00904-4
- Overview information on large, publicly available resources that now power target discovery appears in summaries of the UK Biobank.

Leave a comment